Most natural proteins are composed of multiple polypeptide chains. Such proteins are called oligomeric proteins and the individual chains are called subunits. Enzymes, proteins that catalyze chemical reactions, are most commonly composed of two or four identical subunits, thus they form homodimers or homotetramers. One of the main advantages of oligomeric structure is cooperativity between the subunits, which allows better regulation of enzyme activity compared to monomeric enzymes, which consist of only a single polypeptide chain. Cooperativity is based on allosteric communication between active sites where catalysis occurs and additional regulatory (allosteric) sites. The communication is transmitted via allosteric communication pathways and leads to a conformational change of the enzyme in response to the binding of a ligand at one of these sites, which in turn influences enzyme activity.
Despite the advantages of cooperativity, not all enzymes have evolved into cooperative oligomers. The question is whether these would also benefit from cooperativity. The aim of the project COOPERATE is to answer this question for the family of papain-like cysteine peptidases (PLPs). These enzymes cleave peptide bonds between amino acid residues in proteins. They are mostly monomeric, but the family also includes the tetrameric, non-cooperative dipeptidyl-peptidase I (DPPI). In humans, they play important physiological and pathological roles. Some plant members of the family are also among the most popular peptidases for biotechnological and industrial applications.
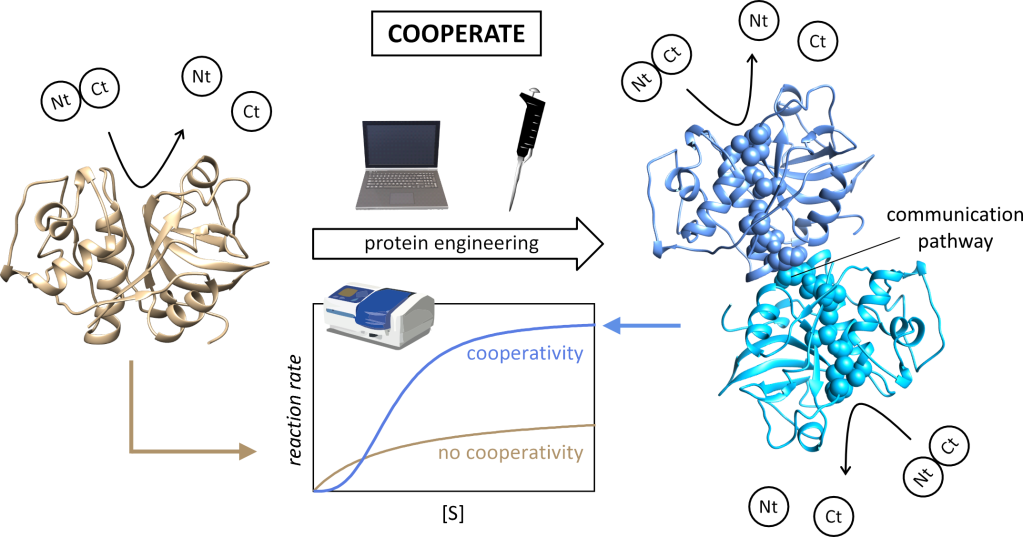
The aim of the project will be achieved by creating oligomeric cooperative variants of these enzymes using two different approaches:
(i) by engineering allosteric communication pathways into human DPPI, which will enable exchange of information between active sites in the form of conformational change, and
(ii) enabling such exchange by engineering homo-oligomeric variants of monomeric PLPs, in which existing communication pathways will be connected at the subunit interface. Human cathepsin K, the best known PLP in terms of allosteric regulation and papain, the most important member of the family for biotechnological purposes, were selected as the primary targets.

Computational model of a hypothetical PLP homodimer.
The project is funded by:


