-
Project COOPERATE
Engineering of cooperative oligomeric peptidases.
PROTEIN
INTERACTIONS & RE-DESIGN

We are a research group at the University of Ljubljana, Faculty of Chemistry and Chemical Technology led by Prof. Dr. Marko Novinec. We specialize in interdisciplinary research at the interface of biochemistry, molecular biology, and synthetic organic chemistry with strong focus on enzymology.
Our research goals are to develop novel tools for protein research and biotechnology, and to discover novel drugs and drug targets.
We are members of Program Group P1-0179.
LATEST DEVELOPMENTS
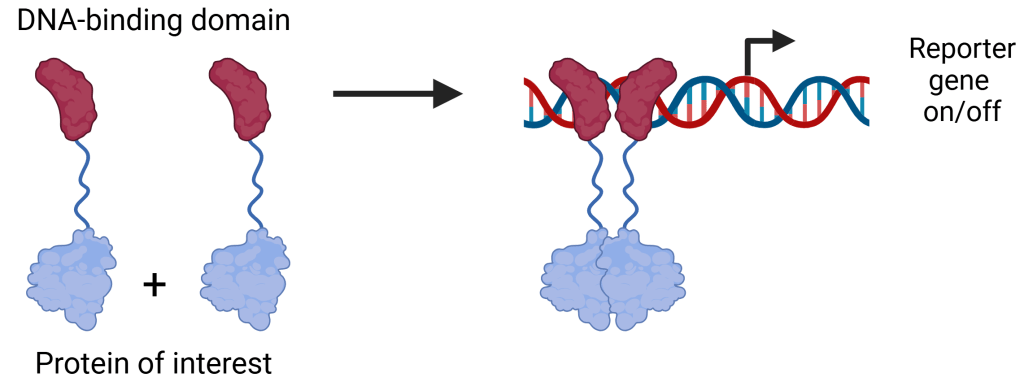
We have received funding from the University of Ljubljana Innovation Fund for further development (to TRL7) and commercialization of our system for the detection, modulation and engineering of homophilic protein-protein interactions in Escherichia coli (codenamed TwinCatcher).
More info about the system will be published soon.
LATEST PUBLICATIONS
RESEARCH PROJECTS
-
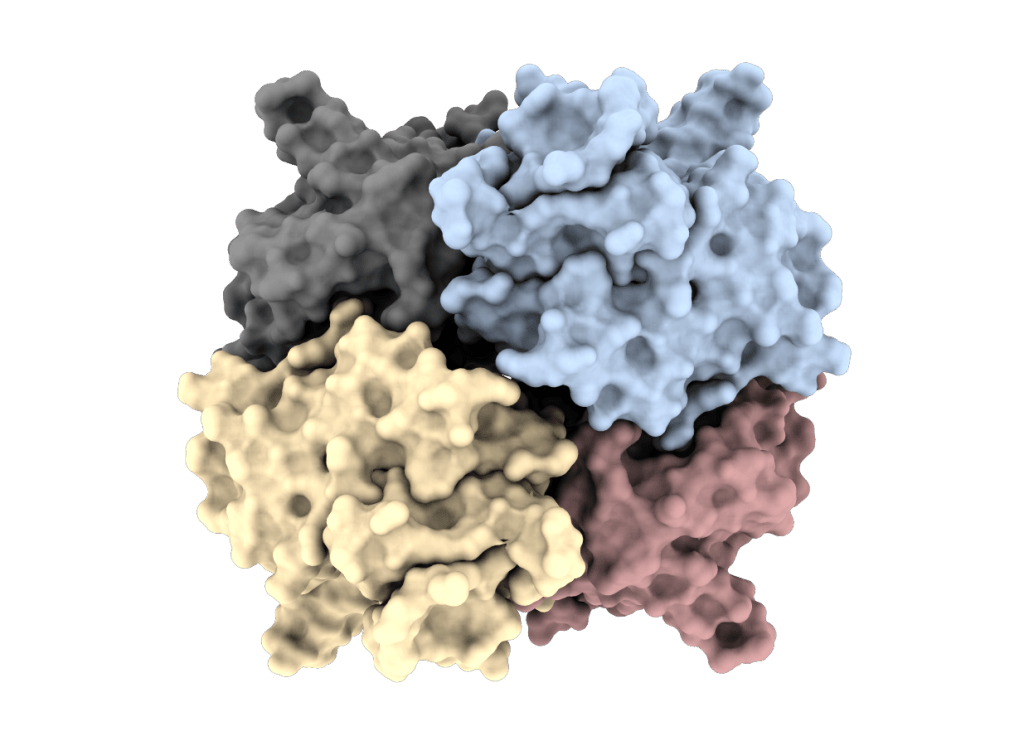
Oligomerization of cathepsin C
Dissecting the molecular structure of a promising drug target.
-
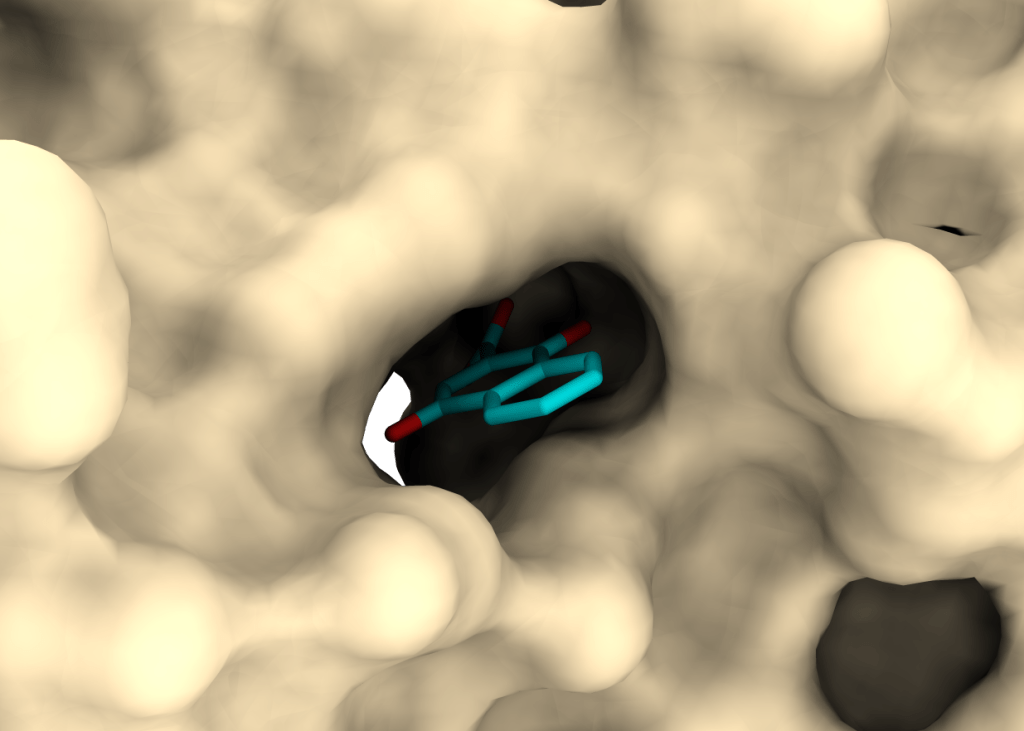
Project PRISM
Discovery of novel antibiotics and drug targets.
-
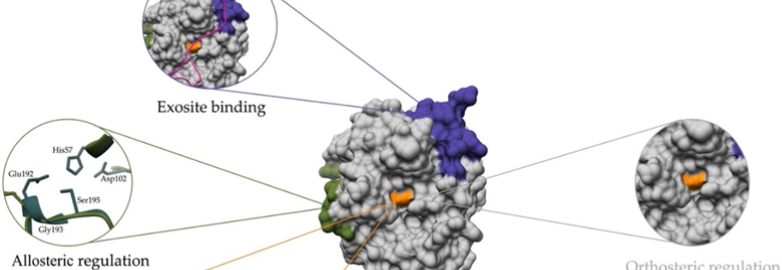
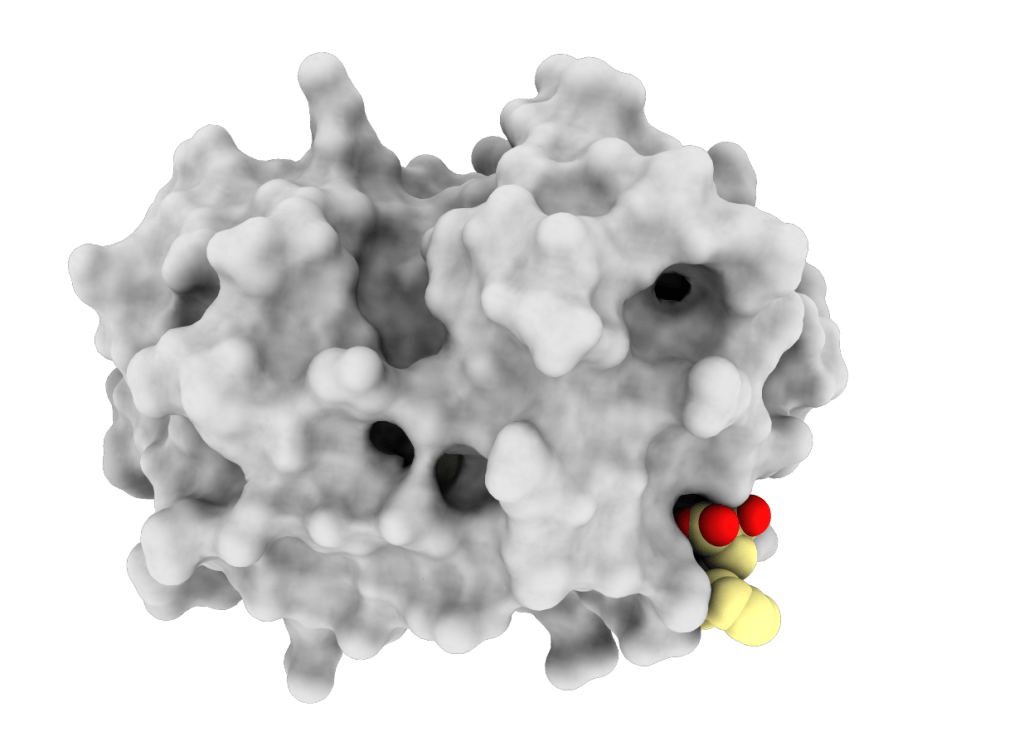
Allostery in Papain-Like Peptidases
Exploring mechanisms of allosteric communication and discovering alternatives to active site-directed inhibitors.
-
COLLABORATIONS
Members of the Ouroboros project team at UL FKKT.
The Ouroboros project investigates the activation of Sirtuin 6 to improve human longevity and health.
-

Contact info:
Dr. Marko Novinec
University of Ljubljana
Faculty of Chemistry & Chemical Techology
Vecna pot 113
SI-1000 Ljubljana
Sloveniaemail to:
marko.novinec@fkkt.uni-lj.si
Designed with WordPress